Overview
In today’s digital world, technologies and software play an increasingly important role in healthcare management - diagnosis, treatment, patient monitoring and Real World Data collection.
This course will help you better understand the regulatory perspectives and standards required for the commercial approval of health products, including risk assessment and mitigation plans. You will gain insights that will support the development, market entry, and effective use of In Vitro Diagnostics (IVDs) and Software as a Medical Device (SaMD)
Course Description & Learning Outcomes
Apply guidelines to perform verification and validation according to requirements
Identify suitable tests and standards for products
Review risk assessment and mitigation plans
List the considerations for software and QC test validation

Schedule
End Date: 28 Apr 2026, Tuesday
Location: 8 College Rd, Duke-NUS Medical School, 169857
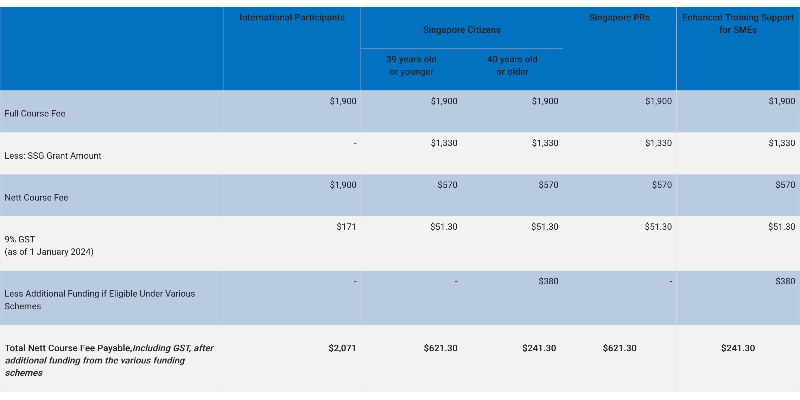
Pricing
Skills Covered
PROFICIENCY LEVEL GUIDE
Beginner: Introduce the subject matter without the need to have any prerequisites.
Proficient: Requires learners to have prior knowledge of the subject.
Expert: Involves advanced and more complex understanding of the subject.
- Optimisation (Proficiency level: Proficient)
- Product Development (Proficiency level: Proficient)
- Product Management (Proficiency level: Proficient)
- Quality assurance (QA) (Proficiency level: Proficient)






