Overview
With the many processes and regulatory requirements for In Vitro Diagnostic (IVD) and Software as Medical Device (SaMD) products, it is a challenging task for any company to ensure compliance. This is a risk to both the public as well as the company, and thus it is crucial to have a system that effectively oversees these processes in maintaining the quality of products.
In this workshop, you will hear from experts discussing key manufacturing requirements, including quality management system standards, and the regulatory steps needed for a company to release a product for sale in regulated markets. This workshop will provide valuable insights into navigating the regulatory landscape for product launch.
Course Description & Learning Outcomes
Gain deeper insights in the processes to ensure the robustness and reliability of manufacturing procedures following the registration and launch of a new product
Learn about the regulatory steps necessary to allow a product to be released successfully into the market
Acquire knowledge on the importance of a well-documented Quality Management System (QMS) and its successful implementation

Schedule
End Date: 19 May 2026, Tuesday
Location: 8 College Rd, Duke-NUS Medical School, 169857
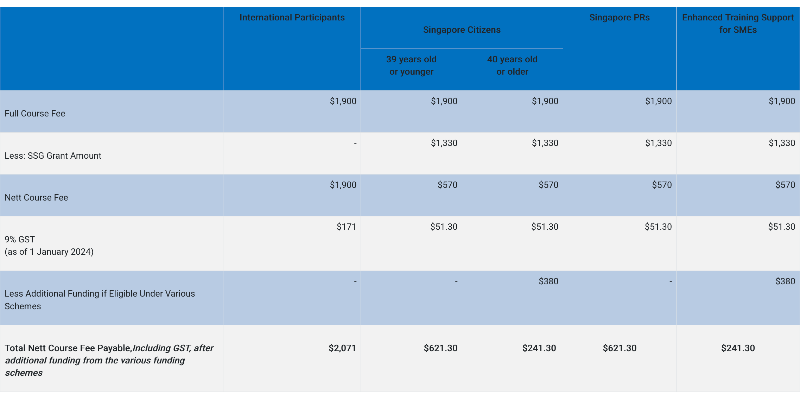
Pricing
Skills Covered
PROFICIENCY LEVEL GUIDE
Beginner: Introduce the subject matter without the need to have any prerequisites.
Proficient: Requires learners to have prior knowledge of the subject.
Expert: Involves advanced and more complex understanding of the subject.
- Product Development (Proficiency level: Proficient)
- Quality assurance (QA) (Proficiency level: Proficient)






